- Premium solutions & products
- Global presence with local support
- Quotation within 24 hours
+61 447 734 654 
- Request a quote
- Become a customer
+61 447 734 654 
All plants need the right amount of food (nutrients) in order to grow. Electrical conductivity (EC) is an essential measurement that indicates the total amount of nutrients available to your plants.

How humidity builds up in Cannabis greenhouses and indoor cannabis cultivation
There are several reasons humidity builds up in a grow room. The two leading factors are transpiration and temperature. Active, living plants transpire water vapor as part of their physiological activity. In a closed environment, this vapor rapidly builds up in the air and may quickly reach saturation. The relative humidity is a factor of both the amount of water vapor in the air and temperature. At lower temperatures, the air can hold less vapor. This drives the relative humidity up. Even a short drop in temperature can cause humidity spikes. When relative humidity reaches 100%, water begins to condense on cooler surfaces, such as railings, pipes or even the plants themselves.
When nutrients dissolve in water, they split into ions. For example, potassium nitrate dissolves into a potassium ion and a nitrate ion. Each ion carries an electrical charge, which creates the potential for electricity to move through a solution. The more ions present, the better the water can conduct electricity.
Plants only absorb nutrients when they are available in an ionic form. That’s why EC is a good indication of the overall strength of your nutrient solution.
Please note: A conductivity measurement doesn’t give you the breakdown of individual nutrients within your solution.
Just as with pH, each type of plant has a preferred EC range within which it can thrive. In general, though, if your EC is very low, your plant probably doesn’t have enough food. On the other hand, if your EC is very high, you run the risk of burning your roots or creating toxic buildups.
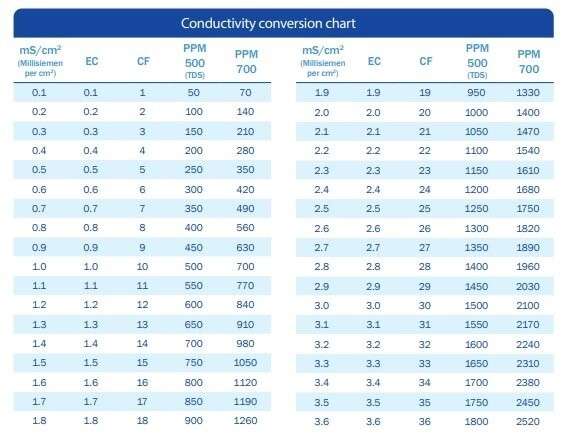
While there’s only one scale for measuring acidity – pH – conductivity isn’t as straightforward. The most common scale for scientific purposes is millisiemens per centimetre (mS/cm2). However, there are at least four other conductivity scales in use.
Common conductivity measurement scales
Always ensure that you know which measurement scale your meter is set on, as well as which scale is being used by your nutrient manufacturer.
• EC (Electrical Conductivity) [1 mS/cm2 = 1 EC]
• PPM (Parts per Million) [EC x 700]
• TDS (Total Dissolved Solids) or DS (Dissolved Salts) or MS (Measured Salts); otherwise known as PPM 500 [EC x 500]
• CF (Conductivity Factor) [EC x 10]
Check out the table below to find out how to convert between these different measurement scales.
Where should you measure conductivity?
There are a few different ways to measure your EC, with the quickest and simplest method being a handheld pen or probe. Here’s where you should think about measuring conductivity and the benefits of each location.
In the nutrient reservoir: This is what you’ll be using to feed your plants, so it’s essential that you check that your reservoir has sufficient nutrient strength.
At the root zone: This is where your plant will be absorbing nutrients. When you measure EC here, this gives you the best indication of the amount of nutrients that are available to your plants.
In the run-off: Measuring here can give you a useful approximation, so long as the percentage of run-off is around 20%.
In the water supply: Unless you are using treated water (via deionised or reverse osmosis), there is a risk of contaminate ions giving an EC value. This could affect your actual EC value and prevent optimal nutrient availability.
Measuring conductivity in soil and soil-less media
There are a few factors to keep in mind when you’re measuring nutrient levels in soil and soil-less media.
Uptake of nutrients by the plant: This will generally decrease the number of nutrient ions and lower the EC. However, your plants may also release other ions that could increase the EC.
The growing medium: Some types of media can release ions - generally by breaking down - which will increase your EC levels. Conversely, some media can sequester nutrients, which will prevent them from being measured.
Reactions between ions: Some ions can react with each other, forming an insoluble complex that’s unable to conduct electricity and therefore unavailable to your plants. A common example of this is the precipitation of calcium sulfate complexes.
Change in water concentration: High evaporation or transpiration rates can result in a more concentrated nutrient solution.
EC changes can happen quickly, which is why it’s so important to measure conductivity regularly. When you do this, you can spot and prevent EC issues as and when they happen.